Data sources and methods
|
Methods
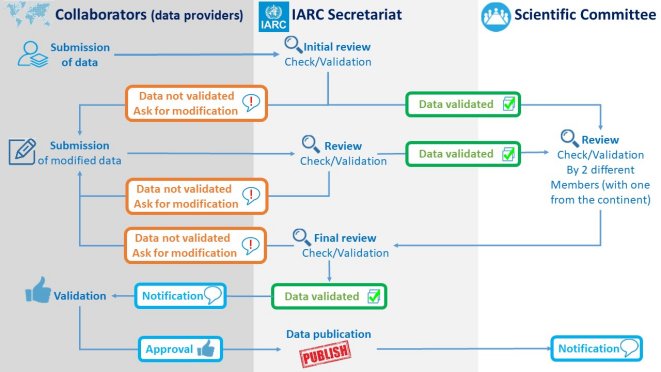
The CanScreen5 project methodology is based on IARC's experience in reporting the status of implementation and performance of breast, cervical and colorectal cancer screening in the 28 Member States of the European Union (Second European Cancer Screening Report, 2017). Collaborators (data providers) and institutions involved in managing and/or supervising cancer screening programmes in different countries are contacted and invited to share qualitative and quantitative information on screening programmes. The current format used for qualitative and quantitative data collection can be downloaded in English and Spanish languages (PDF format): The collaborators will be given password-protected registration, with which they can access the data collection tools and submit the qualitative and quantitative data for the different cancer screening programmes through the CanScreen5 web platform. The data collection tools developed for the Second European Cancer Screening Report are being further improved in consultation with a group of experts (Advisory Board) to make them more broad-based and suitable for different resource settings. The information to be collected is expected to comprise data on national policies, programme characteristics (management, financing, target population, screening practices, quality assurance plan including screening registries, etc.), programme performance (population coverage, screening test performance, diagnostic workup and treatment), and outcomes (detection of disease, predictive values of tests). The project collects data from population-based programmes, opportunistic programmes or pilot/demonstration projects, and population surveys, and categorizes them according to the quality and the reliability of the source. The IARC Secretariat performs the initial review to check the consistency, completeness, and validity of the data. If necessary, collaborators are contacted to complete or modify their submission. Quality checks are carried out to ensure that the collected data meet a defined set of minimum quality criteria. After the data are checked and validated by the IARC Secretariat, the output tables in the form of different process and outcome indicators are automatically generated. The compiled data are shared with the CanScreen5 Scientific Committee to be validated. The individual data provider is contacted again to resolve any queries from the Scientific Committee. Ethical issues, protection of data privacy, and regulatory issues that apply to sharing data are thoroughly reviewed. After the validation process, the newly available country data are added and published on the CanSceen5 web portal. |